Taiwan: 10 Years of the Center for Drug Evaluation (CDE) Health Technology Assessment Division
By Xcenda
HTA QUARTERLY | LATE SUMMER 2018
Taiwan: 10 Years of the Center for Drug Evaluation (CDE) Health Technology Assessment Division
By David Campbell, PharmD, MS
Over 23 years ago, Taiwan underwent a major transformation from fragmented healthcare to a government-run single-payer system. Now, Taiwan’s National Health Insurance Administration (NHIA) provides healthcare coverage to the nation’s whole population of 23.5 million. The universal health coverage scheme provides comprehensive benefits including inpatient and outpatient visits, traditional Chinese medicine, and prescription drugs. To enhance the quality and efficiency of the evaluation of drugs and medical devices, the Center for Drug Evaluation (CDE) was established by the Department of Health (now the Ministry of Health and Welfare, MOHW), and in 2008, a Health Technology Assessment (HTA) division was commissioned to perform the clinical effectiveness and economic assessments of new drugs and devices. The establishment of the HTA division has provided the framework for Taiwan to better deliver quality care while containing costs through efficient use of resources. For the 10th anniversary of the formation of the HTA division, we will review its role within the CDE and the implications for pharmaceutical market access in Taiwan.
The CDE is commissioned by the MOHW and Taiwan Food and Drug Administration (TFDA) to conduct pre-market evaluations of health technologies to assess safety, efficacy, and quality. Based on a similar structure of the United States FDA, applications for marketing approval are submitted to the TFDA through 1 of 3 pathways: a New Drug Application (NDA) for novel therapies; an Abbreviated New Drug Application (ANDA) for generics; or an Over-the-Counter (OTC) Application for non-prescription drugs.
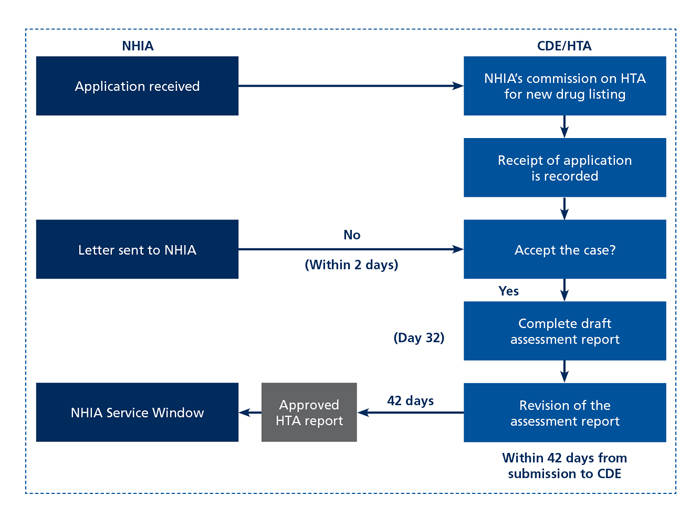
Upon submission of an NDA to the TFDA, an administrative reviewer and review team at the NHIA will be assigned to the application, and a standard review process will take place as outlined in Figure 1. The administrative reviewer will coordinate with the CDE to evaluate chemistry, manufacturing, and controls (CMC), pharmacology, toxicology, PK/PD, and clinical data from the technical dossier of the Drug Master File (DMF). Within the CDE, the HTA division may be asked to conduct comparative clinical effectiveness, economic assessments, and budget impact analyses on new drugs and medical devices of new drug applicants. In this role, the HTA division summarizes available scientific evidence and evaluates the applicability for Taiwan. These roles and responsibilities for the HTA division, outlined in Figure 2, were refined with the second-generation NHIA law implemented in 2013. The assessment reports developed by the HTA division serve as the foundation for the decision-making process on listing new drugs and medical devices and are shared for review and discussion at the Expert Advisory Meeting (EAM). The EAM is usually composed of physicians and at least 1 pharmacist who provide recommendations for NDAs based on their expert opinion. These preliminary recommendations are considered by the Pharmaceutical Benefit and Reimbursement Scheme (PBRS) Joint Committee, which is responsible for final decision making. The CDE / HTA also attend the PBRS Joint Committee meeting to respond to clinical and economic questions. Following a positive decision by the PBRS, the decision is announced and goes into effect upon manufacturer agreement. Contracting agreements may reflect price-volume agreements or other schemes to reduce uncertainty around costs.
Figure 1. Process for Pharmaceutical Listing

Figure 2. Center for Drug Evaluation and HTA Division
Process and Timelines
The influence of the HTA division in PBRS Joint Committee reimbursement decision making seems to be increasing. In 2011, 10 NDAs had accompanying HTA reports; by 2016, the number of NDAs with HTA reports had increased to 22. In an effort to further advance HTA in Taiwan, manufacturers are incentivized to conduct local pharmacoeconomic (PE) analyses to get up to a 10% pricing mark-up. In 2013, the HTA division of the CDE issued guidance for PE assessment methods. While the preferred analytical technique should reflect the purpose of the study, the guidelines generally recommend conducting a cost-effectiveness analysis (CEA) or cost utility analysis (CUA) for drugs with higher therapeutic value. The guidelines recognize that many economic models may originate as international models before adoption to Taiwan. In these instances, the guidelines suggest that Taiwan demographic and epidemiologic characteristics, treatment patterns, financial incentives, and pricing all be carefully reviewed and revised as needed.
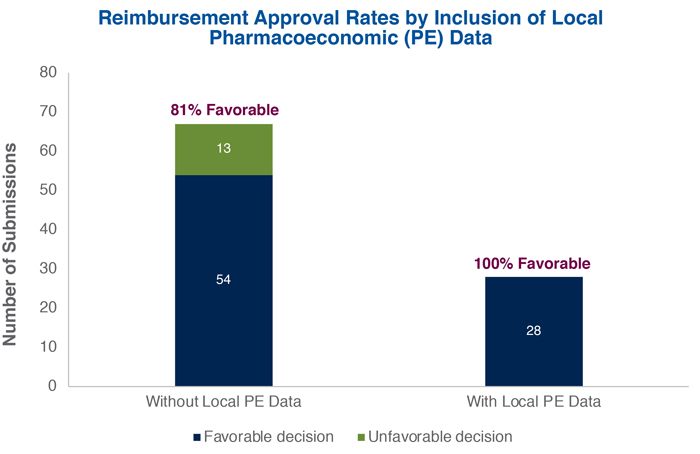
Review of past appraisals suggests there is a correlation between local PE data and reimbursement approval. Over the 6-year period from 2011 to February 2017, 95 NDAs underwent the HTA process, and 86% (82/95) received reimbursement approval. Submissions without local PE analyses achieved a positive recommendation only 81% of the time (54/67), whereas all 28 of the submissions with local PE analyses were approved.
A recent example of the beneficial impact of local PE data on reimbursement is Olumiant® (baricitinib) film-coated tablets for moderate-to-severe rheumatoid arthritis. In this example, the PBRS Joint Committee found the clinical benefit of baricitinib to be similar to other therapies, classifying the drug as schedule 2B and subjecting it to reference pricing (853 yuan/4 mg). However, the presence of quality PE data for Taiwan earned a 4% price premium, and inclusion of Taiwan patients in the clinical trial program earned an additional 10% price premium, raising the reimbursement rate to 972 yuan/4 mg.
If trends continue, HTA and, in particular, local economic evidence will be important for pharmaceutical reimbursement in Taiwan. While only about half of NDAs between 2011 and 2016 with local PE studies received some of the eligible price mark-up for local PE data, all NDAs that included these data were approved. To improve chances of reimbursement approval from the PBRS Joint Committee, manufacturers should prepare local PE data as part of the evidence package.
The article should be referenced as follows:
Campbell D. Taiwan: 10 years of the Center for Drug Evaluation (CDE) health technology assessment division. HTA Quarterly. Late Summer 2018. Sept. 12, 2018.
Sources
- Center for Drug Evaluation, Taiwan. http://www.cde.org.tw/eng/. Accessed July 6, 2018.
- Chang CJ. New Trends of Health Technology Assessment Development and Value Evidence Requirement for Access and Reimbursement in Asia—PE Analysis Practice in Taiwan.
- Chen GT, Chang SC, Chang CJ. New drug reimbursement and pricing policy in Taiwan. Value Health Reg Issues. 2018 May;15:127-132.
- National Health Insurance Drug Payment Project and Payment Standard Joint Meeting, June 21, 2018. https://www.nhi.gov.tw/Content_List.aspx?n=C9AD7A30DC886EFA&topn=3FC7D09599D25979. Accessed August 6, 2018.
